Monitoring Adult Vaccine Use Helps Save Lives


With new vaccines being developed, monitoring their use and acceptability is key.
The world let out a big sigh of relief when we heard the news that COVID-19 vaccines would soon be available to the public. But when the new vaccines came out, lots of misinformation affected acceptability and use. The fact that the vaccines’ development used mRNA technology sounded scary to some because it was the first time vaccines using this technology had been approved. Confidence in vaccine safety and effectiveness has declined in the U.S.
From a public health standpoint, that’s very concerning. Trust between public health institutions and the communities they serve is key to the uptake of life-saving vaccines. At Abt Global, we conduct annual online surveys among pregnant women and health care personnel during the respiratory virus season to support building that trust. Not only do we learn about use of vaccines, but we also ask participants about their knowledge, attitudes, and beliefs on the topic. The surveys are anonymous and offer useful information for real-time public health decisions. We share the findings of these surveys with public health professionals so that they can be responsive to the communities they serve and maintain trust.
The surveys to monitor vaccines began as a partnership with the Centers for Disease Control and Prevention (CDC) to estimate use of flu and tetanus diphtheria pertussis (Tdap) vaccines. Findings from these surveys indicate that flu vaccination among pregnant women has declined in recent years. At the same time, vaccine hesitancy has increased. One out of four pregnant women reported being very hesitant about flu vaccines in 2023 compared with 17 percent in 2022. Pregnant women who are offered vaccination or referred to a place of vaccination during a medical visit are more likely to get vaccinated. This information has been used in vaccine campaigns to encourage pregnant women to speak with their medical providers about the benefits and safety of vaccines during pregnancy.


Since we can modify the online surveys easily, in a matter of weeks after COVID-19 vaccines were authorized for use in the U.S., we worked closely with CDC to add questions about whether respondents received a COVID-19 vaccine, what they thought of the vaccines, and the reasons they did or did not get vaccinated. Four months after vaccines were available, we compiled information from a national sample of people about COVID-19 vaccine uptake and hesitancy, providing CDC with important data for policy decisions. Data showed, for example, that health care personnel working in long-term care facilities and home health care had low vaccination rates compared with those who work in a hospital or outpatient clinic. This is concerning because low vaccination rates among this group mean that already vulnerable patients are at higher risk of infection. In response to this concern, CDC has prioritized health care personnel working in long term care facilities and home health care in communication campaigns to encourage vaccination.
This respiratory virus season, the surveys also will gather information on respiratory syncytial virus (RSV) vaccines. RSV vaccines were authorized for use in the U.S. at the beginning of 2023 and are recommended for older adults and people who are 32 to 36 weeks pregnant. RSV is common and can be very severe among infants. Mothers who get a vaccine while pregnant may pass antibodies to their child to provide protection against RSV during the first few weeks of life.
Every year, Abt uses these online surveys to provide CDC with information to estimate coverage for vaccines for respiratory viruses such as flu and COVID-19. We also summarize data on reasons for getting vaccinated or not getting vaccinated. We learn about where participants get their health information and what information sources they trust. All these data are important because they inform public health institutions about the diverse perspectives that exist in the communities they serve. As perspectives on vaccines in the community evolve, our surveys give public health institutions updated data about vaccine-related knowledge, attitudes, and beliefs. These data can inform efforts to fine-tune messaging around vaccines to promote and protect the public’s health.
For more information on Abt’s influenza research and surveillance work, click here.
Read More
Infectious Diseases & Global Health Security in North America
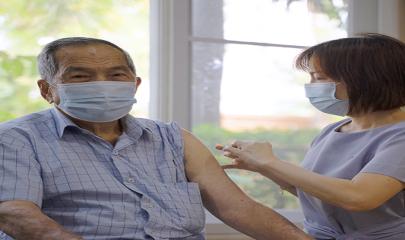
How to Avoid a Respiratory Disease Tripledemic
Getting RSV, COVID-19, and flu vaccinations is the best way to protect yourself and mitigate the odds of an epidemic of all three respiratory diseases at the same time.

Beyond COVID-19: What Global Health Security Will Look Like
As leaders focus on pandemics and global health security, they should look at three related items: other diseases, social determinants of health, and logistics.

A Concise, Health Service Coverage Index for Monitoring Progress Towards Universal Health Coverage
There is a growing international commitment to universal health coverage (UHC), but limited means to determine progress towards that goal. The authors developed a practical index for capturing health service coverage – a critical dimension of UHC -- that was more inclusive than previous methods.The approach moved beyond common indicators of service coverage focused on infectious diseases and maternal and child health, to include information on necessary health inputs. The resulting, balanced, composite index of health service coverage demonstrated promise as a metric, likely to discriminate coverage levels between countries and regions. An important number of service provision indicators were correlated, therefore a reduced set of services performed well as a proxy for the full set of available indicators. This parsimonious index is a start toward simplifying the task of policy-makers monitoring progress on a key domain of universal health coverage.

CDC / CFA Demographic Social Comorbidities Table (DSCT)
The nation’s ability to identify and respond to outbreaks relies on access to high-quality and timely data for disease surveillance.
